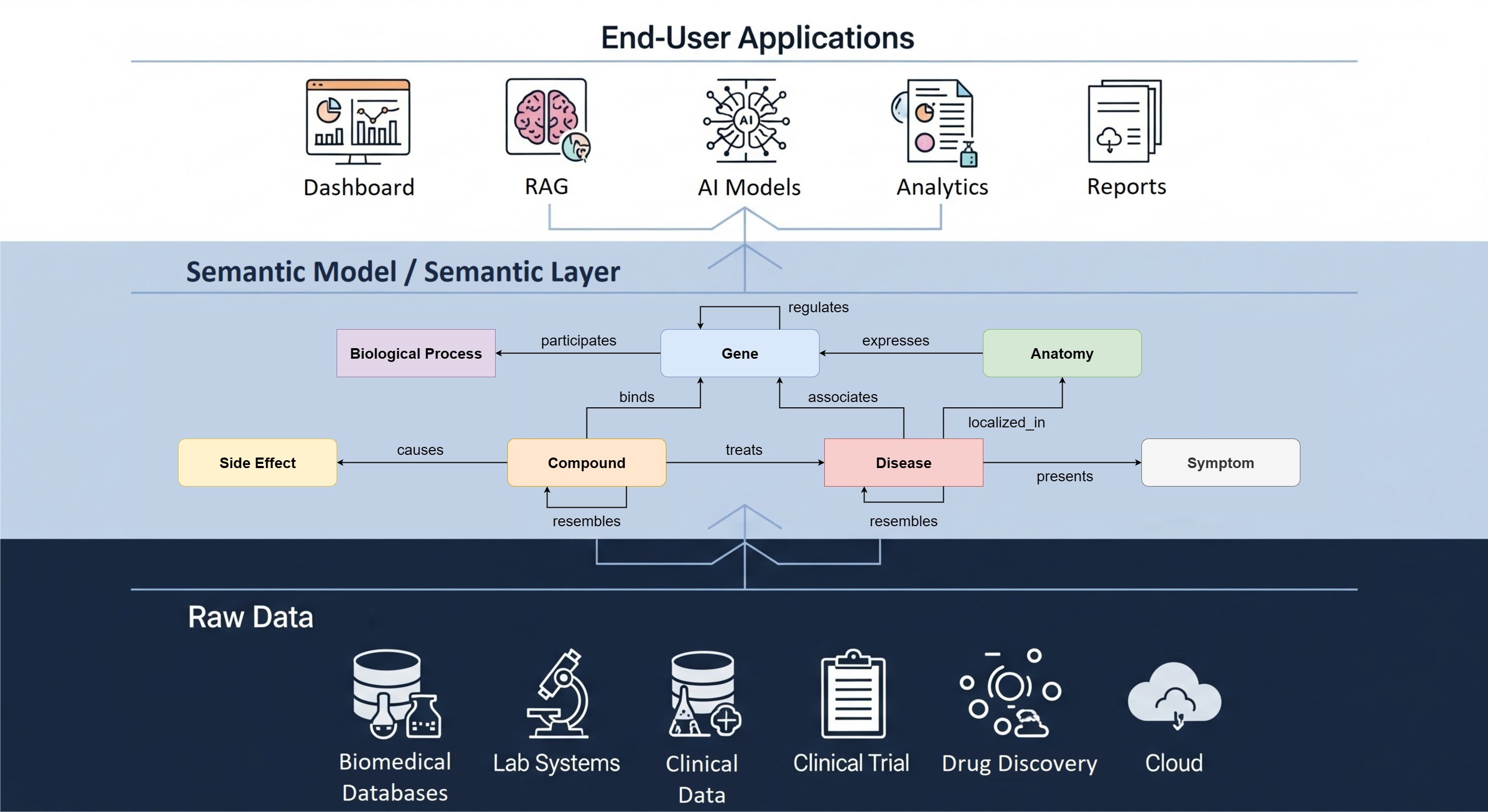
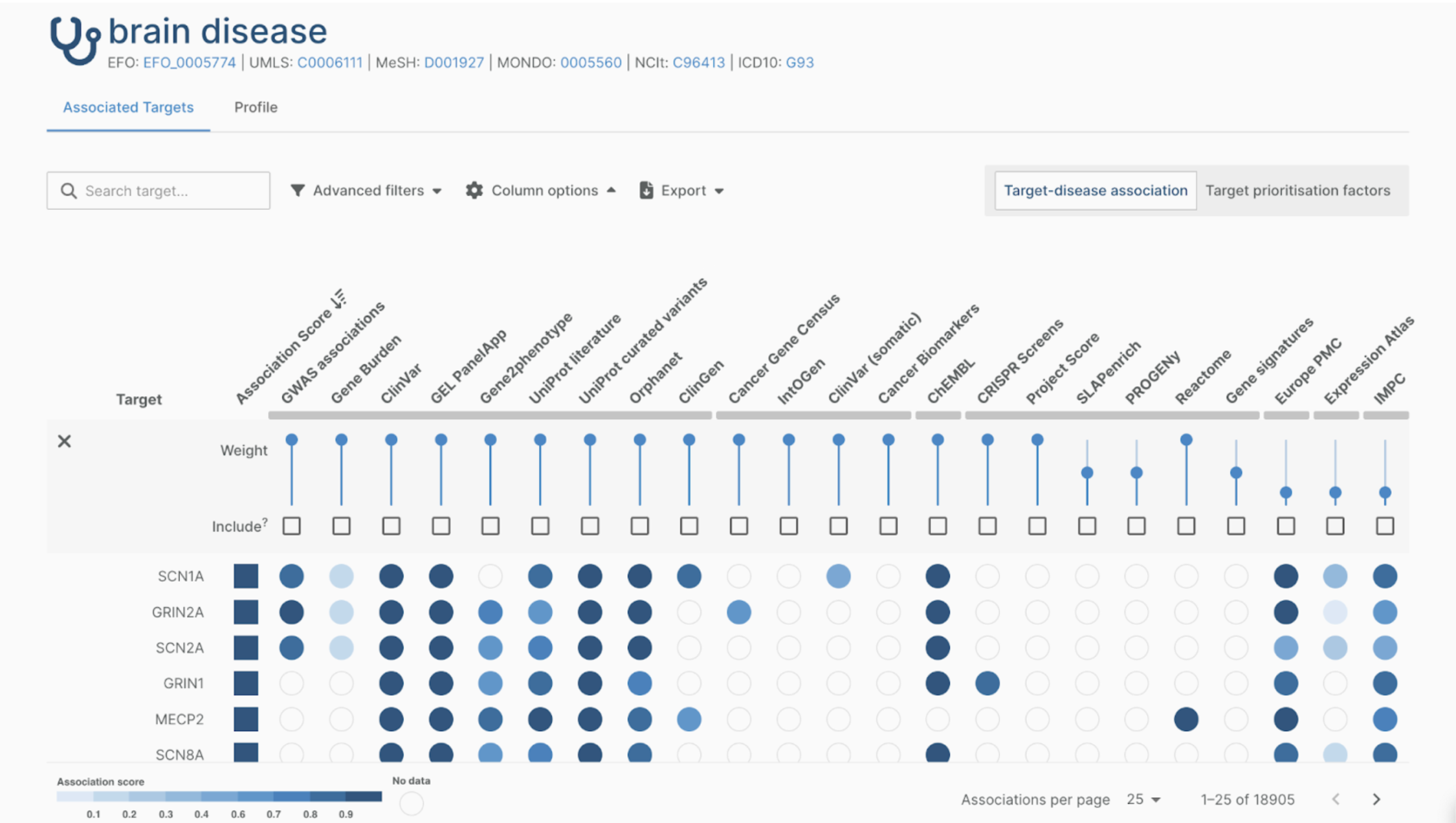
Training and workshops
Our workshops have trained 150+ participants from more than 20 European and US-based life science organizations that have decided to adopt the OMOP CDM and the OHDSI tool suite. We help bring various stakeholders within an organization to the same level of expertise and understanding of the multi-faceted OHDSI ecosystem.